| Previous Chapter | Home Page | Next Chapter |
5. The Thick Mesozoic Atmosphere
Atmospheric Uncertainty
The atmosphere that surrounds our planet is so familiar, so constant, that most people rarely stop to think about it. We check the weather, talk about the heat or rain, and go about our days breathing the air without effort. For many of us, especially those not burdened by illness or poverty, clean air has always been available — an invisible backdrop to our lives. Yet this ever-present mixture of 78% nitrogen and 21% oxygen is anything but ordinary. How did it get here? Why is it made of these particular gases? These are questions that most people — even many scientists — seldom ask. But the air we breathe is not just a given; it’s a puzzle, and one worth solving.
Evolution of the Atmosphere
“The present atmospheres of both Venus and the Earth are thought to have formed through the release of gases from their interiors. The three principal gases that result from outgassing on the Earth are carbon dioxide, nitrogen, and water vapor. Only nitrogen has remained primarily in the atmosphere. Nearly all of the Earth’s outgassed carbon dioxide is locked up in carbonated minerals that form in the oceans and become rocks on the ocean floor. These rocks are eventually subducted into the interior, where the carbon dioxide is released from the rock and recycled back into the atmosphere in volcanic venting. Most of the Earth’s outgassed water now forms the oceans.
John D. Fix, Astronomy: Journey to the Cosmic Frontier, 4th ed., p. 227
Most planetary scientists recognize that volcanic outgassing was the source material for creating the atmospheres of Venus, Earth, and Mars. Since volcanic outgassing consists mostly of water and carbon dioxide, it is not surprising that the atmospheres of Venus and Mars are dominated by carbon dioxide. Yet this raises the question: why is Earth’s atmosphere so different from its neighboring planets? Furthermore, since Earth is the largest and most geologically active terrestrial planet, why does it not also have the thickest atmosphere?
Planetary scientists struggle to explain how Earth went from a roughly 96% carbon dioxide and 4% nitrogen atmosphere, similar to its neighboring planets, to its present-day atmosphere of 78% nitrogen and 21% oxygen. This difficulty arises largely because they incorrectly assume that the thickness of a planet’s atmosphere remains constant — when in reality there is no scientific basis for such an assumption. Even within our own solar system, planetary atmospheres vary enormously, being hundreds, thousands, or even millions of times thicker or thinner from one planet to the next. Moreover, much can happen over the millions and billions of years of a planet’s history. Why should anyone assume Earth’s earlier atmosphere was the same thickness as it is today?
Once the possibility of a much thicker ancient atmosphere is considered, it becomes easy to see how a dense carbon dioxide atmosphere could transition into a relatively thin, predominantly nitrogen atmosphere. If early Earth had an extremely thick atmosphere with a composition similar to present-day Venus and Mars, and then lost nearly all of its carbon dioxide and much of its nitrogen, while gaining oxygen, the result would naturally be a thinner atmosphere of roughly 78% nitrogen and 21% oxygen: the atmosphere Earth has today.
This chapter investigates the implications of Earth’s atmosphere having previously been hundreds of times denser than it is today. Later chapters will explore in greater detail the origins of this thick atmosphere and explain what caused it to thin to its present state.
Understanding Fluids
It may be hard to imagine that the Earth's air could have been so thick that its density was comparable to water. Nevertheless, there is no reason why a gas cannot be compressed to exhibit properties similar to a liquid—in fact, compressing a gas into a liquid is a common industrial process.
For air near the Earth's surface to be so dense, there must have been a substantial amount of overlying atmosphere pressing down on it. Thus, high-density air at ground level serves as evidence of an extremely thick Mesozoic atmosphere.
Unlike water or other liquids, which maintain nearly constant density from top to bottom, a planet's atmospheric density increases as one moves downward from the vacuum of space to the surface. Likewise, atmospheric pressure also rises with depth. Near the planet's surface, both density and pressure are greatest due to the weight of the overlying air compressing the lower layers.
Unlike water or other liquids that have nearly constant density between the top and the bottom, the density of a planet’s atmosphere increases as one travels from the darkness of space downward to the planet’s surface. In addition, there is also an increase in pressure as we move downward towards the surface. Close to the planet’s surface both the atmosphere’s density and the atmospheric pressure is the greatest due to the weight of all of the air above compressing the air at the surface.

Earth's Atmosphere
Above the Clouds
A good approximation of the pressure at the Earth’s surface can be achieved by using the ideal gas law:
P is the pressure in N/m2, V is volume in m3, n is the number of moles, R is the ideal gas constant (8.31 J / mol*K), and T is the absolute temperature given in Kelvin. This equation is typically used for gases in closed containers, but since Earth's atmosphere is not a closed system, we must first apply the substitution:
Where ρ is the density of the atmosphere and M is the average molecular weight of the gas. Making these substitutions gives the equation for calculating the pressure:
\[ P = \frac{ ρ \: R\: T}{M} \]By inserting values of ρ = 660 kg/m³ for atmospheric density, T = 294 K (21°C) for the estimated Mesozoic global temperature, and M = 43.0 g/mol for the molecular weight of the atmosphere, we calculate that approximately 150 million years ago, the Earth's atmospheric pressure near the surface was about 370 atmospheres.
An atmosphere is a unit for pressure. The present sea-level atmosphere is defined as 1.00 atmospheres. Other corresponding measurements of the present sea-level pressure are 1.01 E5 N/m2 (1.01 x 105 N/m2) and 14.7 PSI (pounds per square inch).
A pressure reading taken within a static fluid is an indication of the weight of fluid above that location. So a Mesozoic sea-level atmospheric pressure of 370 atmospheres would indicate that 150 million years ago the Mesozoic atmosphere was 370 times thicker than what it is today.
Before assuming that such a thick atmosphere would have crushed all life on the surface, consider the pressure at the deepest parts of the ocean today. The average ocean depth is 3,790 meters, where the pressure is approximately 380 atmospheres — nearly the same as the calculated pressure at the bottom of the Mesozoic atmosphere. Yet, many species thrive at this depth, and even more exist at much greater depths. Extremely high absolute pressure has no ill effects on modern deep-sea creatures that have evolved in such environments; likewise, the high pressure of the Mesozoic era had no adverse effects on the terrestrial species that lived during that time.
Understanding Pressure

Corals and Tropical Fish
To clear up possible confusion over pressure it may be helpful to recognize the distinction between absolute pressure and pressure difference.
If both the inside and outside of an enclosed container are at the same absolute pressure — regardless of how high that pressure is — there will be no net force on the container's walls. For example, if both the inside and outside of a closed container are at 370 atmospheres, the walls of the container will experience no stress.
In contrast, even a small or moderate difference in pressure across a wall can create a substantial force. For example, imagine a typical window experiencing a pressure difference of just 1/30 of an atmosphere between its inside and outside surfaces. This seemingly small difference would exert a force on the window equivalent to laying it on the ground and having several men stand closely together on its surface. A typical window would shatter under a pressure difference as small as 4 × 10-3 atmospheres.
The distinction between absolute pressure and pressure difference can also be seen in the comparison between submarines and marine life. When a submarine at the water’s surface closes its hatch before diving, the air pressure inside remains at 1.0 atmosphere, the same as the surrounding sea-level air pressure. However, as the submarine descends, the water pressure outside increases dramatically while the air pressure inside remains unchanged. At a depth of 250 meters, the external pressure is about 25 atmospheres, creating a significant pressure difference that pushes inward on the submarine. To withstand these crushing forces, submarines must be built with thick steel hulls.

When submerged, the water outside the submarine is at a pressure several times greater than the pressure of the
air inside. Submarines need a thick steel hull in order to withstand this pressure difference.
Wilhelm Bauer Submarine at Bremerhaven Submarine Museum
In contrast to the rigid submarine, animals that live in the great depths of the ocean experience no ill effects despite existing in an extremely high-pressure environment. Unlike submarines, these animals do not maintain a pressure difference between the interior and exterior of their bodies. Since the pressure inside their bodies matches the surrounding water pressure, there is no net force or strain on them, regardless of how high the absolute pressure may be.
This remains true as long as the animal does not change depth too quickly, thereby altering the external pressure too rapidly. Most people are familiar with the mild discomfort of their ears popping when changing altitude, such as when flying, driving up or down a mountain, or even diving into the deep end of a pool. Scuba divers who ascend too quickly may experience a much more painful and potentially fatal condition known as decompression sickness, or the bends. This effect is not limited to humans — whales and possibly other marine animals can also suffer from the bends if they rise to the surface too quickly.

Water is the main component of sea animals' bodies. Since water is hardly compressible, turtles, fish, and other marine creatures do not need a hard shell and are unaffected by the high pressure of the surrounding water.
Because of these decompression challenges, marine biologists are still developing methods to bring deep-sea creatures to the surface without harming them. To clarify: rapid decompression can cause serious problems for many species, whereas remaining in a stable, high-pressure environment poses no adverse effects.
As human beings living on the Earth's surface, we live at the bottom of a sea of air. At sea level this air produces a pressure on our bodies of 1.0 atm., 1.01 105 Pa, or 14.7 PSI. The area on the face of an average adult's hand is about 0.0116 m2 or 18.0 square inches so there is about 1200 N (270 pounds) of force bearing down on an average adult human hand. Since the pressure is the same for both inside and outside of us, the net forces balance out to zero. Rather than weighing us down, we are indifferent to this force.
Regarding fluid pressure, it does not matter if we are discussing air or water, they are both fluids. The creatures of the Mesozoic era were at the extremely high absolute pressure of 370 atmospheres. Yet just like the deep ocean species of today, this high absolute pressure was on both the inside and outside of their bodies so it produced no stress on their bodies. The extremely high absolute pressure of the thick Mesozoic atmosphere would have had no adverse effect on the dinosaurs.
Light Penetration

To help visualize the thickness of the Mesozoic atmosphere, its density has been compared to that of water. This comparison also helps illustrate how a thick atmosphere could provide buoyancy and what it might feel like to move through such an environment. However, it is important to remember that these two fluids are not identical in all respects. One key difference is how they interact with light — particularly in how far light can penetrate water.
Water is an electric dipole molecule, meaning that while its overall charge is neutral, one side of the molecule carries a positive charge while the opposite side is negative. This uneven charge distribution is responsible for many of water’s unique properties.

Scuba Diver Swimming Past Corals
Water absorbs light from above, so the deeper we go, the darker it gets.
For example, when we use a microwave to heat food, the microwaves interact with water’s dipole molecules, causing them to oscillate rapidly. This oscillation generates heat, which we perceive as thermal energy. Because microwaves can penetrate deep into food before transferring their energy, they enable quick cooking. This ability to absorb electromagnetic energy—including light—is one of water’s defining characteristics.
Light from the Sun does not lose energy as it travels through space to Earth. However, once it penetrates the ocean, much of its energy is absorbed by the water. Most sunlight is absorbed within the first 200 meters of ocean water. Beyond this sunlit layer, known as the epipelagic zone, the light becomes too dim to support plant life. As we descend further, the light continues to fade until complete darkness is reached at around 1,000 meters, beyond which only the deep abyss remains.
If the thick Mesozoic atmosphere had absorbed light in the same way as ocean water, it would have posed a serious problem for dinosaurs and other life forms of that era. However, like today’s atmosphere, the Mesozoic atmosphere likely contained only a small percentage of water vapor relative to other gases. As a result, the vast majority of sunlight would have still passed through to reach the surface and even penetrate the upper layers of ancient oceans.
That said, the Sun and stars of the Mesozoic sky would not have appeared exactly as they do today. Astronomers understand how atmospheric turbulence deflects starlight, causing stars to twinkle. During the Mesozoic era, starlight passing through such a thick atmosphere may have been so distorted by turbulence that individual stars were indistinguishable. Likewise, due to the extreme thickness of the atmosphere, the Mesozoic Sun would likely have appeared far hazier than it does today.
How Solar Radiation Determines a Planet’s Temperature
Without the warmth of the Sun’s illumination, Earth and the other planets would be extremely cold worlds. Fortunately for us, soon after becoming active, our solar nuclear power plant has maintained a nearly constant output of 3.85 × 1026 watts of radiated power. This value is calculated by using an optical thermometer to measure the Sun’s surface temperature at approximately 5,770 K, determining the Sun’s radius to be 3.5 × 108 m, and then applying the Stefan-Boltzmann equation for radiation:
where:
- P is power,
- e is the emissivity factor,
- k is the Stefan-Boltzmann constant, equal to 5.67 × 10⁻⁸ W/m²·K⁴,
- A is the surface area, and
- T is the absolute temperature in Kelvin.
The emissivity factor (e) measures how well a surface absorbs and emits radiation. Depending on factors such as a material’s reflectivity, smoothness, and color, emissivity can range between 0 and 1. Notably, a material's emissivity value is the same for both emission and absorption. This means that if an object readily absorbs radiation when exposed to light, it will just as easily emit that energy as radiation to its surroundings. The Sun, being a nearly perfect emitter and absorber of radiation, has an emissivity value of 1.

Our Solar System
The first four planets — Mercury, Venus, Earth, and Mars — are terrestrial planets. These planets are mostly
solid, with either no atmosphere (Mercury) or a thin to moderately thick one. For a terrestrial planet, the default atmospheric composition is about
96% carbon dioxide and 4% nitrogen. Earth is the exception to this rule.
The last four planets — Jupiter, Saturn, Uranus, and Neptune — are gas giants. These planets are composed almost entirely of hydrogen and helium, and they
are actually much larger and farther away than depicted in this composite image.
Only a small fraction of the light radiating from our Sun is intercepted by the planets of our solar system, yet this light is what warms them. A planet's temperature is primarily determined by its distance from the Sun. The closer a planet is, the greater the intensity of sunlight it receives, making it warmer. The relationship between distance from the Sun and average surface temperature is extreme — ranging from the scorching surface of nearby Mercury to the frigid cold of distant Neptune. Even between Earth and Mars, the drop in average temperature is so significant that Mars' average surface temperature is equivalent to that of Earth's South Pole, the coldest place on the planet.

Relaxing by a Campfire
When we sit around a campfire on a cold night, the radiate heat from the fire will
warm the side facing the fire while our backside is still quite cold.
The equation for intensity is:
\[ I = \frac{P}{A} \] \[ I = \frac{P}{4\:\pi\: R^2} \]where I is intensity, P is power, A is the surface area of a sphere, and R is the distance from the source.
This relationship between distance from a radiant heat source and warmth is familiar to anyone who has sat by a blazing campfire on a cold night. Sitting too close to the fire is uncomfortably hot, while sitting too far away leaves one feeling cold. Just as a person finds a comfortable distance from the fire, Earth happens to be at the right distance from the Sun to maintain a habitable temperature. Also, much like the backside of a person sitting by the campfire, a planet’s "backside" (night side) can also be quite cold.
Our first step in deriving the relationship between a planet’s distance from the Sun and its average temperature is to recognize that all of the radiant power absorbed by a planet is equal to the radiant power that the planet emits back into space.

Light beam coming from a lighthouse.
Light spreads out as it travels away from its source, causing its intensity to diminish.
By first writing the equation for the radiant power gained by the planet and then writing the equation for the radiant power lost by the planet, we can set the two equations equal to each other to find the relationship between temperature and distance.
The radiant power gained by the planet is:
\[ P_{\text{gain}} = P_s \: e\: \left( \frac{A_f}{A_o} \right) \] \[ P_{\text{gain}} = P_s \, e \left( \frac{\pi\: r^2}{4\:\pi\: R^2} \right) \]Where:
- Ps is the power output of the Sun.
- e is the total emissivity of all atmospheric components and the surface of the planet.
- Af is the area of the planet intercepting sunlight.
- Ao is the area of a sphere whose radius is the planet’s distance from the Sun.
- r is the radius of the planet.
- R is the orbital radius of the planet.
The radiant power lost by the planet is:
Where:
- k is the Stefan-Boltzmann constant, equal to 5.67 × 10⁻⁸ W/m²·K⁴,
- Ap is the planet's surface area.
- T is the absolute temperature in Kelvin.
Setting the radiant power lost equal to the radiant power gained and inserting 3.85×1026 watts for the Sun’s radiated power gives the following simple relationship for a planet’s average temperature in Kelvin as a function of its orbital distance from the Sun (with distance in meters):
\[ T = \frac{1.08 \times 10^8}{R^{1/2}} \]Using our new equation we can calculate the average temperature of a planet based on its distance from the Sun. The results are given in the table below.
| Planet | Distance from Sun (Billion Meters) |
Atmosphere | Calculated Temperature Average or Range (Kelvin) |
Actual Temperature Average or Range (Kelvin) |
|---|---|---|---|---|
| Mercury | 57.9 | No Atmosphere | 449 | 103 - 623 |
| Venus | 108 | Extremely Thick | 329 | 753 |
| Earth | 150 | Moderate | 279 | 285 |
| Moon | 150 | No atmosphere | 279 | 110 - 390 |
| Mars | 228 | Thin Atmosphere | 226 | 210 |
| Jupiter | 778 | Gas Planet | 122 | 123 |
| Saturn | 1430 | Gas Planet | 90 | 93 |
| Uranus | 2870 | Gas Planet | 64 | 63 |
| Neptune | 4500 | Gas Planet | 51 | 53 |
Six of the nine major solar system objects show a good match between the calculated and actual temperatures values. The Earth is about fifteen degrees warmer than what the equation predicts while Mars is about fifteen degrees cooler. The Jovian gas planets consisting of Jupiter, Saturn, Uranus, and Neptune show excellent agreement between the calculated and measured values. Of the three remaining objects in the table, Mercury and the Moon are grouped together as the ‘no atmosphere’ category of objects whose temperature varies considerably depending on whether it is day or night, and finally there is the mysterious Venus that is in a category by itself. Let us address the ‘no atmosphere’ category of Mercury and the Moon first.

The derivation of the temperature-distance equation assumes that the Sun's radiation is uniformly distributed around a planet or moon, allowing it to evenly radiate thermal energy back into space. This assumption holds for the Jovian gas planets and the thin-atmosphere planets, Earth and Mars. However, Mercury and the Moon lack atmospheres and do not fit this model. Without an atmosphere, the Sun-facing side becomes extremely hot, while the dark side remains cold.
With no atmosphere, all of the Sun’s radiation directly heats the surface, but at night, there is nothing to retain this warmth, causing rapid temperature drops. In addition to extreme day-night temperature differences, planets and moons also exhibit significant temperature variation between equatorial and polar regions. Near the equator, sunlight strikes the surface more directly, delivering maximum intensity, while near the poles, it arrives at a steep angle and spreads over a larger area, reducing its intensity. As a result, polar regions are much colder than the lower latitudes near the equator.

These temperature differences, whether based on day and night cycles or latitude, are most extreme for objects with no atmosphere and less pronounced for planets with at least some atmosphere. For Mercury and the Moon, the absence of an atmosphere causes such extreme temperature swings between night and day that listing these extremes is more meaningful than providing an average. In contrast, for planets with relatively thin atmospheres, such as Earth and Mars, it makes more sense to use an average temperature while acknowledging temperature variations. On both Earth and Mars, atmospheric temperatures fluctuate between day and night, and there are also significant differences between the high temperatures near the equator and the lower temperatures of the Polar Regions.
The Mesozoic Paleoclimate Paradox

During the Mesozoic era, a remarkably homogeneous flora of tropical and temperate plant species covered the Earth. Plants such as ferns, laurels, palm trees, and magnolias, which could not withstand freezing temperatures, thrived as far as 70 degrees north and south latitude. Many of the same plants that existed near the equator also flourished in the Polar Regions. Alongside this plant life, early crocodiles, dinosaur footprints, and fossilized bones have been found at these high latitudes. Furthermore, for nearly the entire Mesozoic era, geologists have found no evidence of glaciation near the poles, indicating that there was no ice at the poles throughout this period.
For decades, paleoclimatologists have struggled to explain the Mesozoic paleoclimate paradox. When their climate models matched the vegetation of the lower and middle latitudes, they were too cold at higher latitudes. When they adjusted the models to match the warm temperatures of the Polar Regions, the projections depicted an unrealistically hot, sauna-like climate for the rest of the planet, conflicting with geological evidence. Their frustration is evident when they claim that the geological evidence, rather than their simulation models, must be wrong. None of the paleoclimate computer models have come close to replicating the mild, balmy global climate of the Mesozoic era.
The reason these climate models fail is that paleoclimatologists incorrectly assume that the Mesozoic atmosphere was the same thickness as it is today. As stated earlier, temperature variations across a planet — whether between day and night or across different latitudes — depend on the thickness of its atmosphere. Planets or moons with no atmosphere experience the most extreme temperature differences. Planets with relatively thin atmospheres, such as present-day Earth and Mars, still have temperature variations but to a lesser extent. In contrast, planets with extremely thick atmospheres, such as Venus and the Mesozoic Earth, experience almost no temperature differences due to latitude, seasonal sunlight changes, or day-night cycles.

At the equator, the Sun's rays shine nearly directly down on the surface, heating this region more than any other. The air above the surface absorbs this heat and expands. This hot, low-density air becomes more buoyant than the surrounding air, causing it to rise to a higher altitude. As it ascends, it displaces the air above it, while nearby air masses are drawn toward the equator to replace the rising air. Once these new air masses reach the equator, they, too, heat up and ascend, continuing the cycle. This process forms a continuous conveyor belt of air flowing in a circular pattern.
Once convection currents are established, they efficiently transport heat from warm to cold regions. In an ideal scenario, the hot air rising at the equator would travel all the way to the cold poles, but Earth's present atmosphere is too thin to support such elongated convection cells. Instead, the equatorial air descends at around 30 degrees latitude before returning to the equator. Below is a diagram produced by NASA illustrating the idealized three-cell circulation pattern per hemisphere.
To complete the process, each hemisphere has two additional convection cells: one at the mid-latitudes and another at the high polar latitudes. The polar convection cell rotates in the same direction as the strong convection cell near the equator, while the middle convection cell, acting like a middle gear, rotates in the opposite direction. This opposing rotation creates constantly changing weather patterns in the mid-latitudes. While the three-cell system does transfer heat from the equator to the poles, it is far less effective than the single-cell system of a thick atmosphere.
Besides transporting heat from warm to cooler locations, moving air also plays a crucial role in carrying moisture. The amount of moisture air can hold depends on its temperature, with warm air holding much more than cold air. Air near the ground is warmest and can hold the most moisture, but if it rises — whether through convection currents or by being forced over a mountain—its temperature drops, causing it to lose moisture as rain.
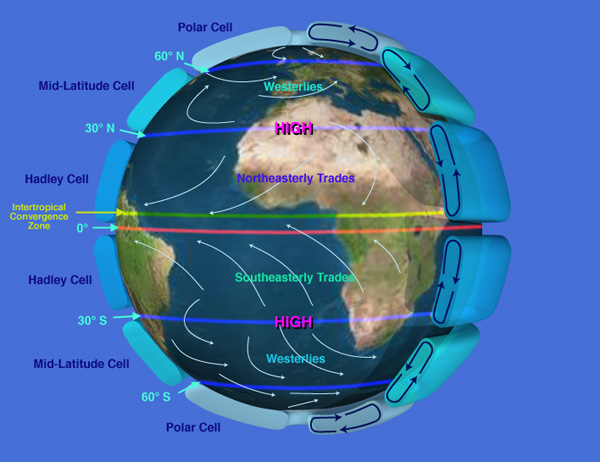
NASA diagram showing the idealized three convection cells per hemisphere circulating pattern of the atmosphere.
Near the equator, where air is constantly rising, rainfall is nearly continuous. Relief from the downpour comes only with the seasonal shift between wet and dry periods. Unlike the hot summers and cold winters of middle latitudes, equatorial regions experience seasons defined by rainfall. Without the Earth's axial tilt, these areas would have year-round rain. However, because of this tilt, the boundary where northern and southern convection cells meet shifts slightly north or south. When this boundary moves overhead, it rains; when it moves away, dry conditions prevail.
Regardless of whether air rises due to equatorial heating or being forced over mountains, the result is the same: rain. As air rises, it loses moisture, becoming extremely dry. This dry air then acts like a sponge, absorbing moisture from the land it moves over. When a moist air mass climbs a mountain range, heavy rain falls on one side, while a desert forms on the other. Similarly, air that loses moisture near the equator descends around 30 degrees latitude, now depleted of water. As it travels back toward the equator, it absorbs moisture from the land, creating deserts.
Hadley Cells — the two dominant convection patterns near the equator — are responsible for placing the world's great deserts between 20 and 35 degrees latitude, both north and south. Refer to the image of Earth to see the brown regions marking these vast deserts.
Deserts are often the best locations for finding dinosaur fossils, yet the animal and plant fossils discovered there typically suggest a moderately humid forest rather than a desert. One possible explanation for this paradox is that continents may have shifted to higher or lower latitudes over time, altering regional climates. Although continental plates usually move only a few centimeters per year, over hundreds of millions of years, this slow movement can significantly change a continent’s position and climate. Another possibility is that rising sea levels sometimes create inland seas, which could increase humidity in surrounding areas. However, even after considering these factors, several large deserts, such as Africa’s Sahara, still present an undeniable climate paradox. The question remains: how can paleontologists find fossils of ancient ferns and other evidence of a once-moist environment in the middle of today’s dry, sandy landscapes?

Idealized One Convection Cells Per Hemisphere Atmospheric Circulating Pattern
To find the answer, we look at Venus, the only planet that closely models Earth's thick Mesozoic atmosphere. Venus’ atmosphere is 91 times thicker than Earth’s, and the Earth’s Mesozoic atmosphere was several hundred times thicker than today's relatively thin atmosphere. Both atmospheres share the characteristic of uniform surface temperature, regardless of latitude, with the equator’s temperature on Venus only slightly higher than at the poles, similar to Mesozoic Earth.
A primary reason for this uniformity on Venus is its highly efficient atmospheric convection system, which evenly distributes solar radiation. With such a thick atmosphere, Venus has one convection cell per hemisphere, transporting heat from the equator to the poles. This one-cell system is much more effective than Earth’s current three-cell system in redistributing heat from lower to higher latitudes. Similarly, Earth’s much thicker Mesozoic atmosphere would have supported a one-cell convection system, a system that is far more effective in transferring heat from the equator to the poles.
The Mesozoic one-cell system had two major effects on global climate: 1) it redistributed heat more efficiently, creating a nearly uniform temperature across Earth’s surface, and 2) it distributed moisture more evenly. Unlike today, where breaks occur near the 30° and 60° latitudes, the lower air currents in the Mesozoic atmosphere traveled all the way from the poles to the equator, resulting in more evenly distributed rainfall.
Besides temperature and rainfall, another important aspect about climate is wind.
Wind gets its energy from the Sun. The Sun heats the surface of a terrestrial planet, and most of this thermal energy determines the planet's surface atmospheric temperature. If the heating is uneven some of the solar energy will contribute to generating wind. Wind is also influenced by the planet’s rotation and convection patterns that establish themselves so as to move warm air to cooler regions. Wind tends to be stronger in areas with greater temperature contrasts, such as coastal regions, mountain passes, and regions with significant seasonal changes.

Sailboats use the energy of wind
So, would a thicker atmosphere on Earth create a windier or less windy climate than today? To answer this, let’s compare Mars, with its thin atmosphere, to Venus, with its thick atmosphere. Mars is known for massive dust storms that can obscure its surface for weeks or months. Venus also has high-speed winds, but they behave differently — while its upper atmosphere experiences extreme winds, surface winds reach no more than a few miles per hour. Since surface winds are more relevant to our question, this comparison suggests a general rule: the thicker the atmosphere, the weaker the winds at the surface. Let’s explore why this is the case.
As stated earlier, the Sun warms a planet’s surface unevenly. Lower latitudes receive the most intense radiation, the poles receive less, and the side facing away from the Sun gets none at all. This uneven heating creates pressure gradients, while a planet’s rotation deflects winds and influences their direction. Geographic features like mountains can channel or block winds, making some regions windier than others. Coastlines create local sea breezes due to differences in how land and water absorb heat.
On Mars, the atmosphere is so thin that surface heating and cooling are nearly as extreme as on Mercury or the Moon—both of which lack atmospheres. These temperature differences create strong regional pressure variations, resulting in Mars’ intense winds.
On Venus, the opposite occurs. Its thick atmosphere acts like an ocean current, with strong convection that slowly and evenly distributes heat around the planet.
Even though Earth's Mesozoic atmosphere was even thicker than Venus', there was still more wind activity than on Venus. This is because, unlike Venus, Earth had vast oceans, lush forests, lakes, and mountains. These varied surfaces absorbed heat at different rates, creating temperature variations and corresponding air pressure gradients that encouraged air movement. Because the Mesozoic air was so thick there would be no gusting winds, but rather an air movement more like water currents that were usually so light that they would hardly be noticed.
Another key difference is cloud composition. While Earth's clouds consisted of water droplets and ice crystals, Venus' thick cloud layer is made of sulfuric acid. These clouds of sulfuric acid block most of the Sun’s infrared, ultraviolet, and visible radiation from reaching the surface of Venus, and they also block this radiation as it tries to escape out to space. The Mesozoic Earth, like today’s Earth, would have lacked sulfuric acid clouds because atmospheric sulfur was removed by rainfall. This is yet another way water makes Earth a unique planet.
In summary, there wouldn’t have been much need for a weatherman during the Mesozoic era. Throughout the year, across the entire planet, the weather was remarkably consistent — mostly sunny skies, a light breeze, and a fair chance of afternoon showers.
The Seasons
It may seem that the Thick Atmosphere Solution has resolved all the major puzzles of the Mesozoic climate, yet one remains: if temperatures were nearly uniform across the Earth — day and night, year-round — how did some Mesozoic trees still develop growth rings? To answer this, it is helpful to review what causes Earth's present-day seasons.
Each year, as the Earth orbits the Sun, we experience seasonal changes. These changes occur because the Earth's rotational axis is tilted by 23.5 degrees. This tilt means that during the northern hemisphere’s summer months, the northern latitudes receive more sunlight than average. In addition to experiencing longer daylight hours, the solar radiation is more intense because the midday Sun is more directly overhead.
It is important to note that while the northern hemisphere experiences summer, the southern hemisphere undergoes winter. Six months later, the situation is reversed—when it is winter in the northern hemisphere, it is summer in the southern hemisphere. It may seem unusual to those of us in the northern hemisphere, but ‘down under,’ December, January, and February are the hottest months of the year.

Diagram Showing How the Tilt of the Earth Produces the Seasons
Generally, seasonal extremes in sunlight and temperature become more pronounced the farther one moves from the equator. In low latitudes near the equator, there is only a slight change in temperature and sunlight throughout the year, and cold winters do not occur. As mentioned earlier, seasons in lower latitudes are defined more by dry and rainy periods rather than temperature fluctuations.
Moving from lower latitudes toward the highest latitudes, temperature and sunlight levels begin to vary dramatically with the seasons. In high latitudes, the transition from summer to winter brings not only extreme cold but also drastic changes in daylight. During winter, the Sun remains low on the horizon, casting long shadows throughout the day, and at the highest latitudes — north of the Arctic Circle or south of the Antarctic Circle — there are winter ‘days’ when the Sun never rises above the horizon.
On present-day Earth, the amount of sunlight a location receives strongly correlates with its temperature — greater solar radiation generally means a hotter climate. However, during the Mesozoic era, this relationship was much weaker due to the highly efficient one-cell convection system, which redistributed heat far more effectively than today’s three-cell-per-hemisphere system. This redistribution minimized temperature differences across the globe, leading to relatively uniform temperatures despite seasonal variations in solar radiation.
On present-day Earth, the amount of sunlight a location receives strongly correlates with its temperature — greater solar radiation generally means a hotter climate. However, during the Mesozoic era, this relationship was much weaker due to the highly efficient one-cell convection system, which redistributed heat far more effectively than today’s three-cell-per-hemisphere system. This redistribution minimized temperature differences across the globe, leading to relatively uniform temperatures despite seasonal variations in solar radiation.
The reason Mesozoic trees developed growth rings similar to those of present-day trees is that a tree’s growth is based on the amount of sunlight it receives rather than on temperature; during the Mesozoic era, the fact that the atmosphere was much thicker made no difference to the seasonal variation in sunlight.
External Links / References
The Earth's Atmosphere
Ideal Gas Law
- Ideal Gas Law - Hyperphysics
- What is the ideal gas law? - Khan Academy
- IDEAL GASES AND THE IDEAL GAS LAW - Jim Clark
Absolute Pressure and Pressure Difference
The Oceans
- Ocean Light penetration - National Ocean Service
- Shedding LIght on Light in the Ocean - Woods Hole Oceanographic Institution
Unique Properties of Water
Mesozoic Paleoclimatology
- Plant Fossils of West Virginia - Monte Hieb
- Climate Model: Data Discrepancies - P. E. Olsen and D. V. Kent
- Geologic Time - Allen Collins, Robert Guralnick and Brian R. Speer
- Climate Factors that Affected the Life of the Mesozoic - Harold Levin
Atmospheric Circulation
Solar Radiation
- Solar Radiation Basics - US Department of Energy
- Types of Solar Radiation - Oriol Planas
- What is Solar Radiation? - IERE
- Solar Insolation
Digging for Dinosaurs
- The Fabulous Chinese Fossils - FOSSIL MALL
- For Fossil Hunters, Gobi is no Desert - NEW YORK TIMES
- 'Mesozoic Cow' Rises from the Sahara Desert - NPR
